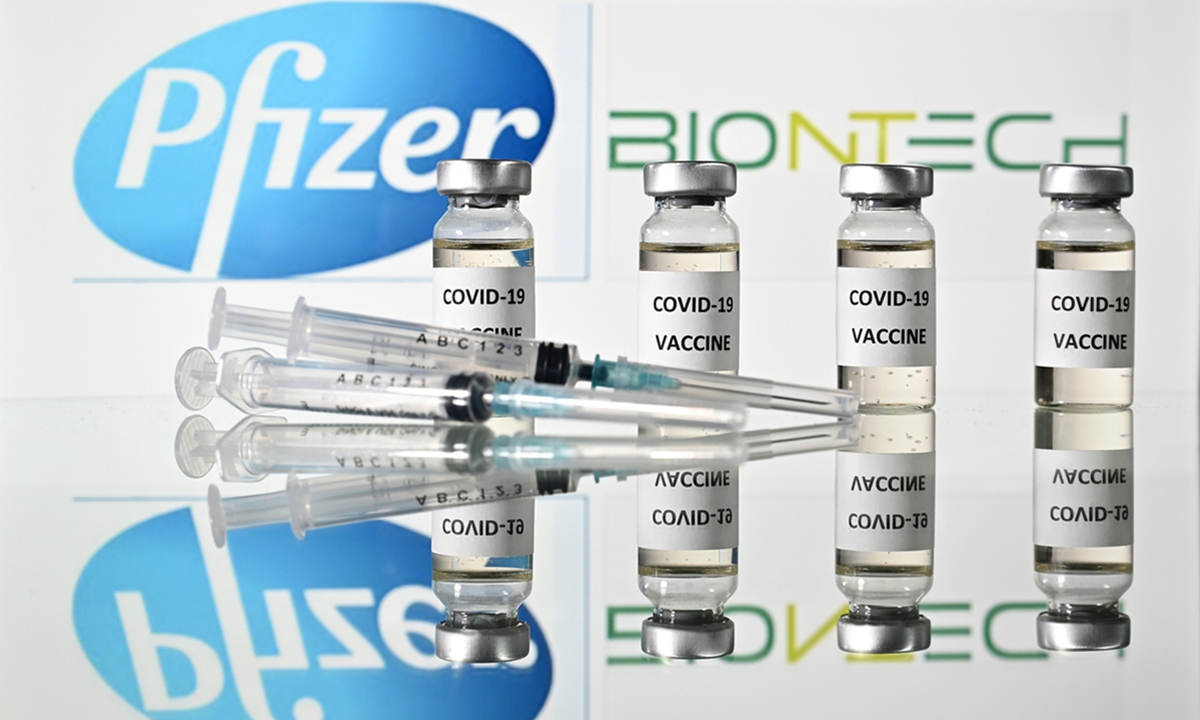
An illustration shows vials of a COVID-19 vaccine and syringes with the logos of US pharmaceutical company Pfizer and German partner BioNTech. Photo: AFP
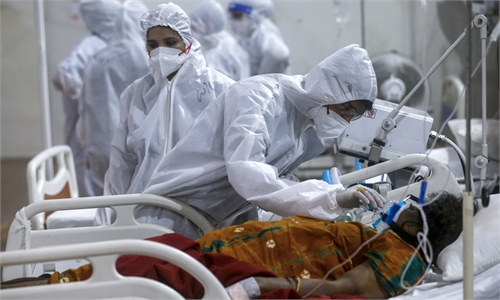
The US on Wednesday authorized the use of boosters of Pfizer's COVID-19 vaccine for people aged over 65, as well as adults at high risk of severe disease and those in high-exposure jobs.
The announcement means a significant part of the population, amounting to tens of millions of Americans, are now eligible for a third shot six months after their second.
"Today"s action demonstrates that science and the currently available data continue to guide the FDA's decision-making for COVID-19 vaccines during this pandemic," said Janet Woodcock, acting head of the Food and Drug Administration, in a statement.
The decision was expected and came after an independent expert panel convened by the regulatory agency last week voted in favor of recommending the move.
The panel, however, rejected an initial plan by the White House to fully approve Pfizer boosters to everyone aged 16 and over, in what amounted to a rare rebuke of President Joe Biden's administration.
The group of vaccinologists, infectious disease specialists and epidemiologists concluded that the benefit-risk balance differed for younger people, especially young males who are more susceptible to myocarditis.
Pfizer COVID-19 boosters are being debated by a separate body of experts convened by the Centers for Disease Control and Prevention (CDC), which may recommend further specifics about recipients.
For example, if obesity is considered as putting a person "at high risk of severe COVID-19," that definition would cover more than 42 percent of the US population aged over 20.
The CDC may also have to define which workplaces and other settings might lead to "frequent institutional or occupational exposure to SARS-CoV-2."
For its part, the FDA indicated this would cover "health care workers, teachers and day care staff, grocery workers and those in homeless shelters or prisons, among others."
The FDA's emergency use authorization applies to those aged 18 and up for the high risk of severe disease and high-exposure categories.
It also only applies to Pfizer's vaccine.
Recipients of Moderna and Johnson & Johnson, the other US-authorized vaccines, will now await news for when they, too, might become eligible for another shot.
AFP